 Energy and
Science Projects
Energy and
Science ProjectsFor Students
 Energy and
Science Projects
Energy and
Science ProjectsElectricity is "created" when certain chemicals react together. We use chemically- made electricity to power many machines from flashlights to a watch or sometimes a car. Yes, there are cars that run on electricity! The devices that store electricity are called batteries. Electricity can also be used to produce chemical changes.
Water is a simple chemical made from two gases -- hydrogen and oxygen. Every molecule of water has two atoms of hydrogen for every atom of oxygen. H2O is the chemical formula for a molecule of water.
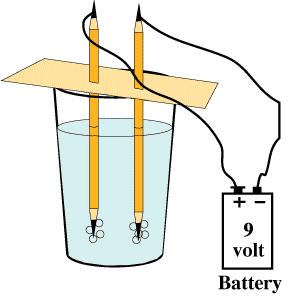
If an electrical current is passed through water between electrodes (the positive and minus poles of a battery), the water is split into its two parts: oxygen and hydrogen. This process is called electrolysis and is used in industry in many ways, such as making metals like aluminum. If one of the electrodes is a metal, it will become covered or plated with any metal in the solution. This is how objects are silverplated.
Try This!
You can use electricity to split water into its two gases -- oxygen and hydrogen.
 What You'll
Need
What You'll
NeedAs the electricity from the battery passes through and between the electrodes (the pencils), the water splits into hydrogen and oxygen, which collect as very tiny bubbles around each pencil tip.
| This Energy Education Project comes from the California Energy Commission | |
|---|---|
 |
 |
| Return to the CEC Science Projects Page | Return to the CEC Energy Quest Home Page |